 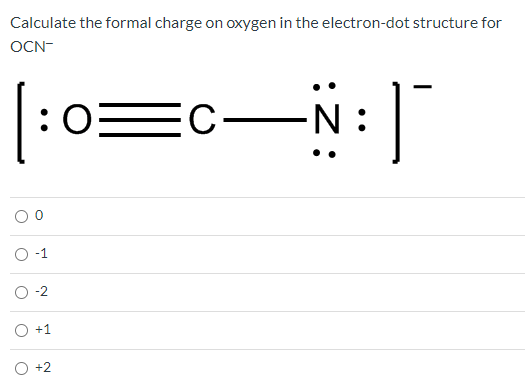 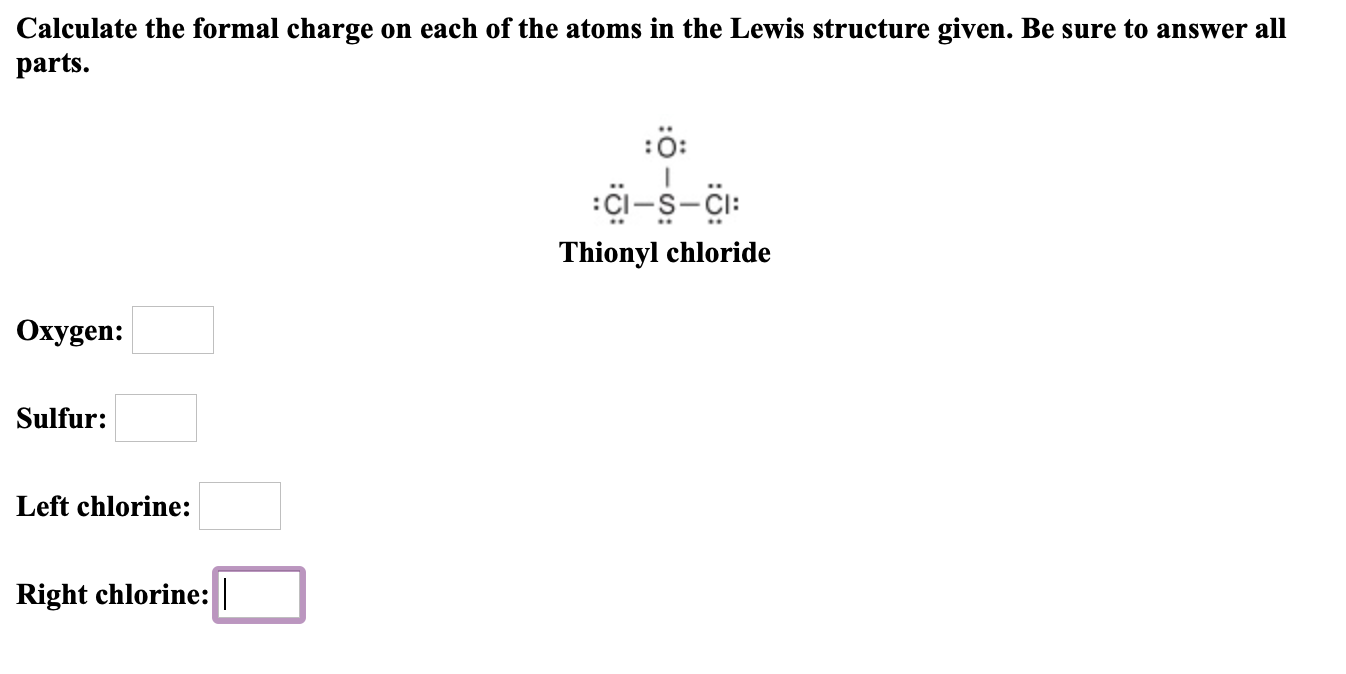
It also gives us a quick estimate of reactivity.Īs an example, second period elements do not exceed the Octet Rule, so you know your structure is invalid if you have 14 electrons on an oxygen atom.īoron compounds often do not have complete octets on boron (for example $\ce$ - 8 electrons, formal charge 2-, oxidation number -2. Another way of saying this is that formal charge results when we take the number of valence electrons of a neutral atom, subtract the nonbonding. The Octet Rule (and the related 18 electron rule for some transition metal compounds) serves to give us some idea of whether the structure proposed meets quantum mechanical requirements for valid structures. The formal charge of an atom in a molecule is the hypothetical charge the atom would have if we could redistribute the electrons in the bonds evenly between the atoms. We can calculate an atom's formal charge using the equation FC VE - LPE - (BE), where VE the number of valence electrons on the free atom, LPE the number of lone pair electrons on the atom in the molecule, and BE the number of bonding (shared) electrons around the atom in the molecule. Formal charge on an atom is calculated as the number of valence electrons in a neutral and isolated atom minus the number of electrons present in its bonded. 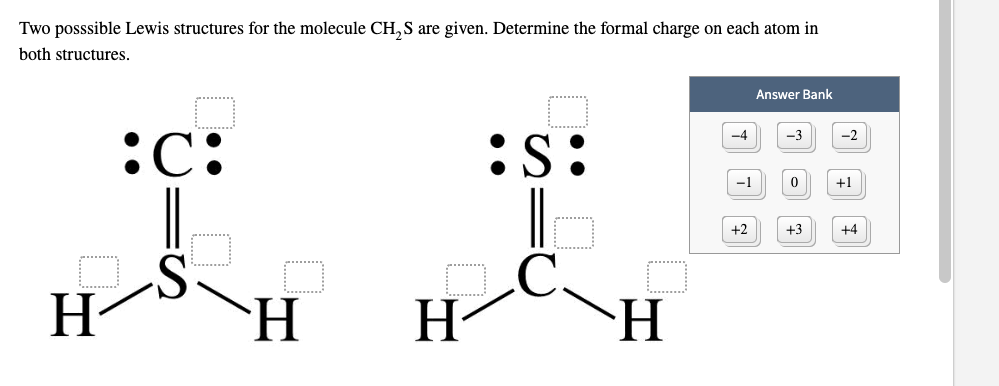
We want to keep track of electrons because it gives us an idea of what type of chemical behavior might be attributable to an atom or molecule. What is the most likely ionization energy and atomic radius for silicon. Calculate the formal charges on the atoms in each and use the results to choose the. As Jan hinted in the comments, there are multiple electron bookkeeping methods which serve different uses. SF4 lewis structure comprises one sulfur and four fluorine atoms. The sum of the formal charges of all atoms in a molecule must be zero the sum of the formal charges in an ion should equal the charge of the ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
